Gene Therapy for Type 1 Diabetes: Preclinical Promise
Despite eclectic ways of delivering insulin to control blood glucose level in people with type 1 diabetes (T1D), no approach precisely replicates what happens in the body. Gene therapy may hold the answer.
T1D is usually autoimmune, with inherited risk factors such as certain HLA haplotypes contributing to, but not directly causing, the condition. A clever use of gene therapy is to commandeer liver cells to step in for the pancreatic beta cells that autoimmunity destroys.
MILESTONES IN HISTORY

One of the most classic stories of modern medicine concerns T1D: discovery of insulin at the University of Toronto in the early 1920s. Young surgeon Frederick Banting and medical student Charles Best were grudgingly given lab space and ten beagles to conduct their famous experiments that identified “isletin”.
In the summer of 1921, the hormone kept alive Marjorie, a dog whose pancreas had been removed, and in January 1922, it saved the first patient, 14-year-old Leonard Thompson.
Human insulin was the first drug produced using recombinant DNA technology. From shocking new biotechnology circa 1982, human insulin is today an item easily picked up at a drug store. But effectively using human insulin requires frequent monitoring and timing of injections, and even the most careful schedule sometimes can’t prevent long-term effects of uncontrolled blood glucose levels.
The prototype insulin pump dates to 1963; a wearable form came a decade later. Many people use pumps today to provide insulin in a manner more like a pancreas.
 Thousands of islet transplants — the cell clusters that include the beta cells — have been done since 1966. But patients need lifelong immunosuppression, and many eventually require supplemental insulin. Each year about 1,300 people in the US receive pancreas transplants, but organs are in short supply and powerful immunosuppressant therapy necessary.
Thousands of islet transplants — the cell clusters that include the beta cells — have been done since 1966. But patients need lifelong immunosuppression, and many eventually require supplemental insulin. Each year about 1,300 people in the US receive pancreas transplants, but organs are in short supply and powerful immunosuppressant therapy necessary.
STEM CELLS AND GENE THERAPY
The challenge in treating T1D is not just to provide insulin, but to quell the autoimmune response against the antigens that festoon beta cells. The disease isn’t due to a mutation in the gene that encodes proinsulin, which is naturally cleaved to release the peptide insulin. Because cell surfaces are what attracts the misdirected immune response, a cell-based approach would need to provide cells that do not have surfaces resembling beta cells. Stem cells are receiving lots of attention. Clinicaltrials.gov lists 88 entries under “type 1 diabetes and stem cells.” Most are hematopoietic or mesenchymal, including one “status unknown” trial using menstrual blood.
Where there are stem cell offerings, hype may follow, or at least overenthusiastic word choices. One company’s home page opens with “Breakthrough Stem Cell Technology,” with the subhead “Transforming the Lives of Patients with Diabetes.” The technology: human embryonic stem cells whose daughter cells differentiate as beta cells once slipped under a patient’s skin. I’m not sure how that would be blind to the immune system. It takes a few levels of clicking to get to the “Product is in the Clinic” part of the website, which implies that doctors are using it. They’re not. FDA gave the go-ahead for an initial clinical trial in 2014; “in the clinic” and “in clinical trials” are not at all the same thing. WebMD reported on the cells a few weeks ago, leading with “Patient testing has started on a stem cell treatment that could offer a cure for type 1 diabetes.” Accurate, but perhaps premature.
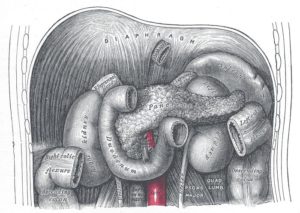 Differentiated cells, such as other types of pancreas cells and liver cells, can be stimulated to secrete insulin, from their own proinsulin genes. But they tend to do so at a continuous low level, not in the complex pattern that is in sync with an organism’s metabolism.
Differentiated cells, such as other types of pancreas cells and liver cells, can be stimulated to secrete insulin, from their own proinsulin genes. But they tend to do so at a continuous low level, not in the complex pattern that is in sync with an organism’s metabolism.
In contrast to the 88 hits for stem cells and T1D, “type 1 diabetes and gene therapy” brings only 19 hits, 18 of which make no sense. The 19th is for a trial of gene therapy targeting p53 (a tumor suppressor gene) in people with diabetes and liver cancer. That trial hasn’t started yet.
Gene therapy for T1D has been in the works for two decades preclinically, at a number of labs. Last week, Hans Sollinger, MD., Ph.D, the Folkert O. Belzer Professor of Surgery in the Section of Kidney and Pancreas Surgery, Division of Transplantation at the University of Wisconsin told me about his work. I’ve written so much about gene therapy for rare diseases that I haven’t given much thought to using it for common conditions. Tackling an autoimmune condition seems more complex than replacing an enzyme or clotting factor.
DNA MINICIRCLES RETOOL LIVER CELLS
Dr. Sollinger and his colleagues call the gene therapy strategy “minimalist”. Instead of making more beta cells, which could still evoke an immune response, they and others retool other cell types that lie beneath the radar of immune surveillance to do what beta cells do, by including the genetic controls with an introduced gene — not just revving up the proinsulin gene already there.
Hepatocytes – the most abundant type of liver cell – are a good stand-in for pancreatic beta cells for a few reasons:
• the liver and pancreas come from the same embryo layer
• hepatocyte surfaces won’t spike autoimmunity
• the cells normally crank out many proteins
• few cells likely need be altered to get a response
• hepatocytes naturally sense and respond to glucose level changes, using a transporter protein that takes up glucose and an enzyme that metabolizes it. Only a few cell types do both, and the liver cells do it best.
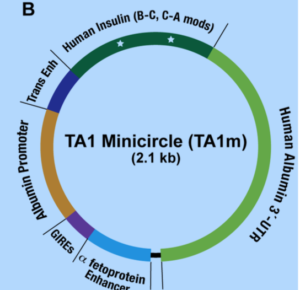
The researchers used rats given a drug that destroys beta cells, inducing T1D. A paper in PLOS One describes their “gene therapy-based glucose-regulated hepatic insulin production.” It’s a human proinsulin gene that’s part of a minicircle of DNA that includes splice sites for an enzyme to trim proinsulin, the albumin promoter that normally starts transcription, and controls that ease and amplify insulin production. The controls ensure that the minicircles work only in hepatocytes, and that they synthesize enough insulin, frequently enough, to lower blood sugar sufficiently to have clinical impact. The minicircles form a complex with the cell’s DNA but don’t insert into chromosomes, so they can’t activate cancer-causing genes, as retroviruses have done in some gene therapy trials.
The approach works: within 45 minutes blood glucose levels of the rats evened out, and stayed that way. “We have animals well beyond 1 year with perfect regulation — all with a simple tail vein injection,” Dr. Sollinger said. That success is being echoed elsewhere. Mathias Elsner, MD, of Hannover Medical School in Germany and his group have successfully used a similar somatic gene therapy approach using lentivirus as a vector, and Peter Thule, MD, from Emory University, has had similar results in mice.
Only about 2% of the liver’s cells need take up and express the transgene, researchers estimate, for a clinical effect, but replication in a large animal model is essential to better predict if that will be enough in humans. PharmaCyte Biotech Inc. will be doing such studies on a “bio-artificial pancreas” consisting of encapsulated human liver cells that secrete insulin. The approach is based on gene therapy in autoimmune diabetic rats developed over two decades by Ann Simpson, PhD, from the University of Technology in Sydney.
The need for better treatment of T1D is compelling — it affects 1 to 1.5 million people in the US, many of them children, at a cost of $8-14 billion a year. Gene therapy can offer cost-effectiveness with few treatments, no immunosuppression or surgery necessary, and is possible for all patients. The next step is dogs, back where it all started. I hope that T1D will one day follow in the footsteps of the gene therapies that will likely be FDA-approved within the next year.

[…] Source: Gene Therapy for Type 1 Diabetes: Preclinical Promise […]
[…] Gene Therapy for Type 1 Diabetes: Preclinical Promise – PLoS Blogs (blog) […]
thank for sharing , such a nice information.