Cow Embryos Reveal New Type of Chromosome Chimera
I’ve often wondered what happens between the time an egg is fertilized and the time the ball of cells that it becomes nestles into the uterine lining. It’s a period that we know very little about, a black box of developmental biology, because observing or doing anything to even the earliest human embryos is frowned upon.
Every time I write the following paragraph in new editions of my textbooks, I try to picture what can go wrong during those first cell divisions:
“The birth of a healthy baby is against the odds. Of every 100 eggs exposed to sperm, 84 are fertilized. Of these 84, 69 implant in the uterus, 42 survive one week or longer, 37 survive 6 weeks or longer, and only 31 are born alive. Of the fertilized ova that do not survive, about half have chromosomal abnormalities that cause problems too severe for development to proceed.”
 A lot of what goes wrong happens in the first few days. One early glitch is if both genders are not represented. Despite current discussion over what exactly constitutes being male or female, it is a genetic fact that a fertilized ovum requires one genome from a female and one from a male. An unfertilized egg (it technically isn’t an ovum until it’s fertilized) that doubles its genome doesn’t survive, and an egg that loses its own genome but gets two from sperm forms a tissue mishmash called a hydatidiform mole (aka molar pregnancy) that may or may not contain any embryo parts.
A lot of what goes wrong happens in the first few days. One early glitch is if both genders are not represented. Despite current discussion over what exactly constitutes being male or female, it is a genetic fact that a fertilized ovum requires one genome from a female and one from a male. An unfertilized egg (it technically isn’t an ovum until it’s fertilized) that doubles its genome doesn’t survive, and an egg that loses its own genome but gets two from sperm forms a tissue mishmash called a hydatidiform mole (aka molar pregnancy) that may or may not contain any embryo parts.
Something else can happen, something unexpected, and it’s reported in the May issue of Genome Research: an embryo can have some cells whose two genomes come from the female parent, and some cells whose genomes come from the male. It’s a little like a line of male-female square dance partners separating and coming back in a way that includes some same-sex partners. Joris R. Vermeesch, Ph.D., professor of molecular cytogenetics and genome research at the University of Leuven and colleagues cleverly paired technologies to deduce the existence and origins of these “heterogoneic” chimeric embryos, which means “of differential parental origin.”
 The researchers examined 23 early embryos resulting from in vitro fertilization and culture in “synthetic oviductal fluid” from 8 Belgian blue cows and 2 Holstein-Friesian bulls. A few cell divisions generated 160 blastocysts, the hollow fluid-filled balls of cells that meander down the uterine tubes en route to implantation in the uterus. The researchers sampled representative blastomeres (single cells), and subjected them to whole genome amplification and genotyping of 770,000 single nucleotide polymorphism (SNP) landmarks, as they had the 8 cows and 2 bulls.
The researchers examined 23 early embryos resulting from in vitro fertilization and culture in “synthetic oviductal fluid” from 8 Belgian blue cows and 2 Holstein-Friesian bulls. A few cell divisions generated 160 blastocysts, the hollow fluid-filled balls of cells that meander down the uterine tubes en route to implantation in the uterus. The researchers sampled representative blastomeres (single cells), and subjected them to whole genome amplification and genotyping of 770,000 single nucleotide polymorphism (SNP) landmarks, as they had the 8 cows and 2 bulls.
The investigators developed a technique, called haplarithmisis, which teams detection of copy number variants (numbers of copies of each chromosome or of specific DNA sequences) with SNP analysis. CNV analysis just says that there are equal numbers of each of the 23 human chromosome types – it is blind to the number of chromosome sets. Adding SNP genotyping reveals which parent contributed which chromosome set. Knowing the parents can distinguish a fertilization glitch from the mysterious parental doubling of heterogoneisis.
In the past, chimeric embryos were thought to arise only from those fertilization glitches: two sperm barrel into an egg; two very early embryos glom together; an errant polar body left after meiosis pierces a fertilized egg. But the new work reveals that weird fertilization isn’t necessary to generate the different-parented embryos – it can just happen following a normal egg-meets-sperm event. The male genome doubles in some cells, the female in others, and perhaps some cells remain normal diploid from two parents. “This is a novel fundamental insight into the origin of chimerism, a very rare condition in humans which can lead to birth defects,” Dr. Vermeesh said.
Only 6 of the 23 embryos had all normal cells. “A staggering 39% of embryos (9/23) contain blastomeres with full genome anomalies” — extra or missing chromosome sets. Two-thirds of these had extra chromosome sets, 30 percent had losses, and 3% had all DNA from one parent.
The SNP patterns indicated which parents had contributed which genomes to the embryos. All sorts of anomalies turned up. One 8-celled embryo had two cells that had “scattered fragments of the paternal genome and no maternal inherited DNA.” Apparently DNA from the sperm had gone on replicating even though the egg’s genome had left town. Another normally-fertilized egg had fractured into three cell lines: one chromosomally normal, one with a double dose of the maternal genome, and the other with a double dose of the paternal genome. Dividing by three had been known only in cancer cells and in cells of certain insects and crustaceans. But apparently it can happen after a normal fertilization too.
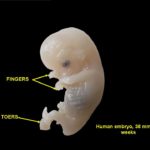 “Miracle” isn’t in the scientific lexicon. But with everything that has to go right, chromosomally speaking, to launch an embryo on the journey of prenatal development, I find it amazing that as many as 31 healthy babies are born for every 100 fertilized eggs.
“Miracle” isn’t in the scientific lexicon. But with everything that has to go right, chromosomally speaking, to launch an embryo on the journey of prenatal development, I find it amazing that as many as 31 healthy babies are born for every 100 fertilized eggs.
(Opener photo credit: Maaike Catteeuw, Ghent University)

[…] Source: Cow Embryos Reveal New Type of Chromosome Chimera […]