Can Liquid Biopsies Compete with Scopes and Scans in Cancer Diagnosis?
March is National Colorectal Cancer Awareness Month, the perfect time to bring up the value of colonoscopies and mammograms. These procedures may seem old school for old folks, but they save lives – more directly than genetic testing. Scopes and scans saved mine and my husband’s lives.
The Value of Visuals
Larry had been getting frequent colonoscopies to check for polyps. In April 2016, I watched as the ashen roto-rooter doc hurried out of the procedure room with a sample, bellowing “call FedX! STAT!” He then came over to talk to me, pulling the curtains closed around the cubicle where Larry sat on a cot still babbling incoherently from the sedative.
“There’s something there, but I don’t know what it is, and I couldn’t remove it. But I got a bit for a biopsy,” the doctor paused, glancing at Larry with concern. “He’ll need surgery.”
The gastroenterologist didn’t recognize the growth because it wasn’t colon cancer. Larry’s rare cancer was actually in the stump of his long-gone appendix, poking into the doctor’s territory.
A routine mammogram found my recent cancer, and after that I had genetic testing, to guide treatment. With this week’s FDA approval of direct-to-consumer testing for the three Ashkenazi mutations in the two BRCA genes, the sequence will reverse, with people who have mutations moving on to mammography to actually find the cancer. I fear the return of the test (FDA blocked it in 2013) will lead to misunderstanding and unnecessary surgeries, but I’ll tackle that another time. I now know personally that removing body parts that you can see is life-altering, and it won’t vanquish all risk and fear, for cancer cells can lurk in what remains. Identifying a mutation is not a diagnosis.
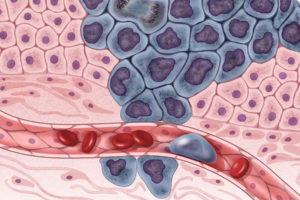
But back to scopes and scans. They alone can’t diagnose cancer, but a savvy physician’s opinion of what they show, I’ve found, is fairly reliable. Tumors must be sampled to spot the telltale changes of cancer: changes in the shape of cells or their nuclei, excess receptors visible with appropriate probes, and comparison to surrounding tissue to reveal invasion. Larry’s sample came out with his colonoscopy; mine was a separate torture.
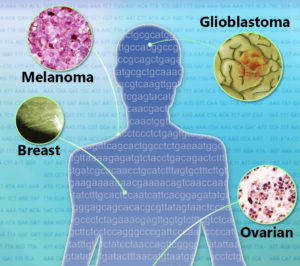
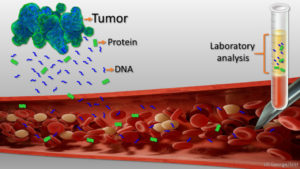
And so the idea of a “liquid biopsy” to find tumor DNA in the bloodstream sounds both tame and tantalizing. But it’s also a tad arrogant, “biopsy” suggesting that it is as diagnostic as examining a slice of tissue riddled with clearly cancerous cells. DNA findings may indeed be more specific than an elevated protein biomarker like prostate-specific antigen (PSA), but the DNA may come from cancer cells that will never go on to cause disease. Liquid biopsy won’t reveal those cells, or even where they are in the body, nor predict which of them will and which won’t go on to divide themselves into a solid tumor. Yet.
Circulating Tumor DNA
A liquid biopsy detects pieces of DNA 150 nucleotides long that come from well-studied oncogenes and tumor suppressor genes – “circulating tumor DNA” (ctDNA). FDA approved the first such test in 2016, to inform treatment choices in patients with metastatic non-small cell lung cancer.
Liquid biopsy can detect recurrence less painfully than a surgical sampling, or spot new mutations conferring drug resistance, or monitor response to treatment. A study published in PLOSOne in 2015 validated the technique – for people with advanced cancer. But a more recent test of two company’s commercially-available liquid biopsies to detect recurrence of metastatic prostate cancer often led to conflicting findings. The tests scrutinize different parts of different genes. Should they really have different results if applied to the same samples?
If liquid biopsy is not reproducible among cancer patients, how informative can it be when used as a screen, which means the general healthy population? Will it be meaningful even among people with common risk factors, such as family history of cancer, liver cirrhosis, or smoking? We don’t know.
A Curious Meeting
Two years ago, I planned to post here on liquid biopsies, but nixed the idea as a series of strange events transpired.
It was an hour before the first plenary session of the day at the American Society of Human Genetics annual meeting in October 2015, and a few days after the PLOSOne article had appeared validating liquid biopsy for cancer patients. A man bearing tee-shirts and the offer of free hot breakfast beckoned, if I’d listen to a lecture on liquid biopsies. Sure! My badge said “media.”
The sponsoring company had recently announced plans to market a test consisting of 96 “driver” (cancer-causing) mutations in 9 cancer genes. At the session they discussed “surveillance” of the “Healthy 100,” folks aged 20 to 71, to estimate prevalence of selected cancers in populations with risk factors but not diagnosed cancer. Liquid biopsy of the “Healthy 100” found four people with mutations indicating cancer – all over 50.
But the unlucky quartet might not actually have had cancer. A paper from 2014 showed that DNA snippets representing cancer genes tend to appear in the circulation as we age, apparently innocuously. So although on a population level the mutations are associated with increased cancer risk, they may not actually ever cause cancer in a particular individual.
I took pages of notes, my skepticism growing as visions of false positives from benign disease and false negatives from not collecting enough DNA or not including enough mutations played in my head. Finally, a noted professor took the stage and put the potential of liquid biopsy into perspective:
“What can it really tell you? It may indicate previously undetected cancer. But it’s not a diagnostic test, and follow-up clinical testing is required to confirm the presence or absence of a specific cancer. A positive result doesn’t definitely mean cancer nor does a negative result indicate not. The aim is to help physicians identify patients at early stages of disease, when cancer is localized, for better treatment options.”
The company planned to market two versions, for “high risk but otherwise healthy” people like the 100, as well as for cancer patients. They’d use a “subscription” service, with quarterly repeat tests for a few hundred dollars a pop, a little like a pricy Netflix.
The CEO had already announced the “launch” in a September 10, 2015 news release: “Early detection is the single most important factor in ensuring successful treatments and improved survival rates.”
Well, not always. And “ensuring” is a red flag where cancer is concerned.
If early detection is “the single most important factor in saving lives,” why are cancers commonly found in autopsies of people who never had symptoms? This is true for cancers of the thyroid and prostate, and for ductal carcinoma in situ (DCIS), the type of breast cancer that I have. And even though diagnoses of DCIS have skyrocketed in tandem to improved resolution in mammography, that hasn’t apparently had much of an impact on catching invasive and metastatic breast cancers, which it should if early detection is predictive. Wouldn’t it be more valuable to know which hints of cancer will actually unfold as disease?
At the end of the session, things got peculiar. I raised my hand to ask questions, identifying myself as a journalist. A stream of “We can’t answer that”s followed. The session ended, and as others filed out, dumping their breakfast trays, company reps and execs rushed over to ask that I send them anything that I intended to write about the session. That’s not done.
But I never wrote the piece – someone had beaten me to it.
Orac is the “nom de blog of a humble surgeon/scientist who has an ego just big enough to delude himself that someone, somewhere might actually give a rodent’s posterior about his copious verbal meanderings,” aka David Gorski.
On October 2, right before the event at the genetics meeting, he’d posted, “Liquid Biopsies For Cancer: Not Ready for Prime Time.” “I fear that this is the recipe for the ultimate in overdiagnosis,” Orac argues. That’s not the same as a false positive, for the test does pick up cancer gene DNA. It’s the interpretation that is at issue, for people who don’t have symptoms.
Thanks to Orac, I discovered that on September 24, two weeks after the company’s news release, they’d received a letter from the FDA challenging the conclusion in a company white paper that liquid biopsy could be used in initial diagnosis. Orac argued that the white paper demonstrated no such thing, but included evidence of efficacy in already-diagnosed individuals. The FDA letter wasn’t mentioned at the breakfast meeting, which might explain the concern over having a reporter in the room.
So now, two years later, I checked the company’s website to discover the fate of liquid biopsy. It’s now festooned with lifestyle-type offerings, as many genetic tests have devolved to recently, like skin products and analysis of such things as IQ, mental health, and “corporate wellness,” which is apparently a thing in some dubious way related to DNA. An online chat quickly confirmed that liquid biopsy is not among the company’s offerings.
But the story isn’t over.
Revisiting Liquid Biopsies
A research report in the February 23 issue of Science opens the next chapter in the continuing saga of liquid biopsy – combining ctDNA detection with protein biomarker levels. They’re familiar in cancer screening: PSA for prostate, CEA for colorectal, CA125 for ovarian. Biomarker testing of healthy populations goes in and out of favor, with the latest take just days ago arguing against PSA testing.
The study in Science by Joshua Cohen from Johns Hopkins and colleagues is elegant, yet it too deploys the technology on people who already have cancer. It’s a little like a student getting answers to last year’s exam and seeing how many questions she would’ve answered correctly.
The new algorithm, CancerSEEK, combines data from 16 cancer genes and 8 biomarkers to determine the risk of developing any of eight solid tumors, then adds 31 more proteins and gender to predict where the cancer is – key information because the same mutations lurk in cells in different body parts. An accompanying Perspective has a nice graphic. NIH funded development of CancerSeek as a potentially “universal” tool that would address several cancers.
The 1005 patients profiled in the study had/have nonmetastatic cancer of the ovary, liver, stomach, pancreas, esophagus, colon, lung, or breast. Results were impressive, finding 99% of the cancers and returning positive results in only 7 of 812 healthy controls. And CancerSEEK was quite good at localizing the cancers.
Even though the investigators conclude that what’s really needed are large, prospective studies that predict, rather than confirm, cancer, they’re looking towards a diagnostic that echoes the multifaceted nature of cancer treatment. In addition to snippets of cancer gene DNA, liquid biopsy of the future might include messenger RNAs, microRNAs, and methylation patterns to capture gene expression, as well as additional metabolites.
Investors are already quite interested in liquid biopsy for the general population, to the tune of a projected $10 billion market, according to one report. But until liquid biopsy becomes meaningful as a cancer screen – that is, for people without symptoms – my advice is to keep going for those unpleasant colonoscopies and mammograms, or start at the appropriate age. Chugging antifreeze and emptying your bowels, or enduring a boob smasher for a few moments, are preferable to cancer. They’re lifesavers!
Here are guidelines for colonoscopy and mammography.


[…] Source: Can Liquid Biopsies Compete with Scopes and Scans in Cancer Diagnosis? […]