Parent Roundtable: Before and After Gene Therapy
I dedicate this post – my 200th here at DNA Science – to the rare disease families I’ve met since starting this blog four years ago. You are all amazing!
As the House of Representatives has just passed the revamped 21st Century Cures Acts and the Senate is expected to pass it soon, I’m reminded of a spellbinding dinner on Sunday night, October 16. It was the eve of the annual “breakthrough summit” for the National Organization for Rare Disorders, and I’d introduced a group of new friends to share their stories of life with a child who has had, or will have, gene therapy.
The first gene therapy may finally receive FDA approval in 2017, perhaps the blindness treatment from Philadelphia-based Spark Therapeutics. The milestone will make headlines, but for thousands of families it will mark just one step on a very long and winding road.

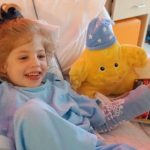
The Scene: Legal Seafoods, near Reagan National Airport. Not everyone at the table knew about the remarkable O’Neill family from South Carolina, who kept 5-year-old Eliza at home, in quarantine, for 726 days awaiting the gene therapy that she received last May. Sanfilippo syndrome type A had already rendered Eliza silent, and she would not have survived her teens. The “very bad disease clogs up her brain and that makes her not learn very well. She’s hyper,” explained healthy older brother Beckham in the “Saving Eliza” video that went viral.
Gene therapy delivers genes aboard viruses, but doesn’t replace mutations. If Eliza caught the same type of virus, she’d make antibodies, disqualifying her from a clinical trial beginning at Nationwide Children’s Hospital in Columbus, Ohio. Hence, the isolation.
Next to Glenn O’Neill sat Beth Frigola-McGinn, whose daughter Ellie is Eliza’s age. Ellie has a mitochondrial disease with a name so impossible to remember– “leukoencephalopathy with brainstem and spinal cord involvement and increased lactate” – that Ellie herself recently launched a social media contest to change it.
As Glenn explained how he and his wife Cara had protected Eliza, enshrouding visiting therapists in masks and gowns, Beth’s eyes widened as her jaw dropped. On her other side, Jocelyn Duff was also transfixed. She’s new to the rare disease community. Would gene therapy halt her 10-year-old daughter Talia’s very rare form of Charcot-Marie-Tooth disease, called CMT4J? Talia, who also has trisomy 21 Down syndrome, is already wheelchair-bound and losing strength in her arms due to the completely unrelated and unexpected CMT4J neuropathy. (I’ll tell her story in a future post.)
Beth’s look of incredulity intensified. “Wait a minute. What? WHAT DID YOU DO?” she finally blurted in a crescendo.
Glenn continued to describe the isolation that his family endured, and minutes later, we all sat back, stunned. Finally, Beth leaned forward.
“I would do exactly what you did,” she whispered, as Jocelyn nodded.
Glenn and Cara are now waiting, watching, and hoping. And things look good.
“Since treatment, we see a new light in Eliza’s eyes – she’s connecting with us in a way we thought was gone forever. She’s working to regain the skills the disease stripped away in the 6½ years it tore through her body,” Glenn told me a few weeks after the dinner. Eliza said, unprompted and appropriately, “mommy” and “daddy” and made it through an hour-long restaurant dinner without a meltdown.
A pessimist might suggest that Eliza knows something important has happened and she’s trying harder, but there’s already measurable evidence of an effect from the gene therapy. Early findings from Abeona Therapeutics on the first three children given the one-time infusion show decline in the biochemical buildup that is the hallmark of the disease, shrinking livers and spleens, and no adverse effects. Safe, so far.
Gene therapy works, but it’s still in clinical trials in the US, none yet approved. The first clinical trial began, in September 1990, at the National Institutes of Health Clinical Center. The European Medicines Agency approved its first gene therapy, Glybera, for a rare inherited pancreatitis, in 2012. But Glybera has encountered problems, such as cost, difficulty of delivery (multiple injections into muscle), and a regulatory morass, obstacles that are not the case at all for the blindness gene therapy. So like most biotechnologies, gene therapy has been a long time in coming. And it’s hit a few much-publicized speedbumps, chronicled in my book about gene therapy.
But thanks to those clinical trials for gene therapies now inching towards publication of phase 3 results, regulatory approval, and ultimately commercialization, more than 200 people with hereditary blindness can now see. People with hemophilia are making clotting factors, and children with certain inborn errors of metabolism, leukodystrophies like adrenoleukodystrophy (ALD), rare neurological conditions such as giant axonal neuropathy (GAN), and immune deficiencies may have longer-lasting alternatives to costly enzyme therapies – or first-time treatments.
Approval of the 21st Century Cures Act would help not only efforts to develop treatments for rare diseases, but fund the Precision Medicine Initiative and the Cancer Moonshot too. Let’s give more children with life-shortening genetic diseases, if not all of them, the same moonshot that Eliza O’Neill has had.
Organizations that are fundraising for children with the diseases mentioned here:
Eliza: Cure Sanfilippo Foundation
Ellie: A Cure for Ellie
Talia: CureCMT4j
 MY NEW BOOK: Human Genetics: The Basics, second edition
MY NEW BOOK: Human Genetics: The Basics, second edition
When Routledge Press asked me to write the lone science book for “The Basics” series in 2009, I was intrigued after churning out textbook tomes for years. I was thrilled when they requested a new edition, out December 13. The six chapters cover the nuts and bolts of DNA science, seen through the lenses of health, diversity, evolution, and even the possible future of humanity.
Pre-order from Routledge or from Amazon.
Thanks for reading DNA Science and a big shout out to PLOS for providing me with this forum.