Genetics in 2016: Breakthrough, Breakdown, and Bridge
I dislike end-of-year “best of” lists, especially the “breakthroughs” that imply scientific discoveries and medical advances emerge from out of nowhere. Often they’re the product of PR machines that select and then catapult certain research findings into the news releases that dictate the headlines.
WHAT MAKES SCIENCE NEWS?
Much of science news is released to journalists ahead of time (embargoed) so that we can investigate background and conduct interviews. This year, the Food and Drug Administration began offering news even earlier to select media outlets. Charles Seife writes in “How the FDA Manipulates the Media” that through the “close-hold embargo” offered to “top-tier media organizations, … FDA cultivates a coterie of journalists whom it keeps in line with threats.”
The rush to report the same stories is why here at the bottom tier, DNA Science seeks the unusual, the research and reflections that others miss or that aren’t promoted. Hence my scintillating coverage of How the Pangolin Got its Scales and frequent focus on families who are living with rare diseases.
 Thanks to PR (and a nasty patent battle), CRISPR was a top 10 contender for Time magazine’s 2016 “person-of-the-year”. CRISPR is perhaps a better buzzword than the zinc finger nucleases and TALENS that are the gene-editors that have been in clinical trials since 2009. And it is an easier technique. Still, I think more deserving is gene therapy, which began clinical trials in 1990, with the first FDA approval likely coming in the new year. (When I pitched that to the top-tiers, all I got was rejection.) So that’s where I begin my list-of-sorts, celebrating a breakthrough, a breakdown, and a bridge of DNA science for 2016.
Thanks to PR (and a nasty patent battle), CRISPR was a top 10 contender for Time magazine’s 2016 “person-of-the-year”. CRISPR is perhaps a better buzzword than the zinc finger nucleases and TALENS that are the gene-editors that have been in clinical trials since 2009. And it is an easier technique. Still, I think more deserving is gene therapy, which began clinical trials in 1990, with the first FDA approval likely coming in the new year. (When I pitched that to the top-tiers, all I got was rejection.) So that’s where I begin my list-of-sorts, celebrating a breakthrough, a breakdown, and a bridge of DNA science for 2016.
BREAKTHROUGHS
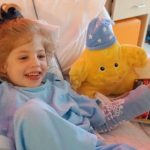
Eliza O’Neill wasn’t the only person to receive gene therapy in 2016, but I choose her to exemplify my measure of a breakthrough: a child who could no longer speak, now singing with her dad, filling in the lyrics to This Old Man. It happened just 6 months post-gene therapy.
The O’Neills had kept Eliza quarantined at home for nearly two years to avoid viral infection that might have disqualified her from the clinical trial for Sanfilippo syndrome type A. Biochemical evidence also suggests the gene therapy is working.
Hannah Sames finally had her gene therapy midsummer this year, for giant axonal neuropathy. It’s too soon to tell whether her new genes are synthesizing gigaxonin, the protein her body can’t make. Meanwhile, Hannah’s working hard to rebuild muscles and help awaken the motor neurons that may rejuvenate them.

Europe is well ahead of us in gene therapy approvals: in 2012 for lipoprotein lipase deficiency and in 2016 for severe combined immune deficiency due to adenosine deaminase deficiency (SCID-ADA). Will the first FDA approvals for gene therapy come in 2017? I think so!
#1 will likely be for the condition that inspired my book about gene therapy, although when I wrote it in 2012 I’d thought approval was just around the corner, so striking is the improvement, even more dramatic than singing a nursery rhyme. At that time, the visual loss was known as LCA2, for Leber congenital amaurosis type 2. Since then it’s become “RPE-65-mediated inherited retinal disease” and the gene therapy, from Spark Therapeutics, dubbed Voretigene neparvovec.
#2 could be hemophilia B. It’s a great candidate for gene therapy because just a slight increase in factor IX level translates into improved blood clotting. A biochemical change heralding a clinical response is the holy grail. Several clinical trials are ongoing at Spark Therapeutics, University Medical Center in Rotterdam, UniQure, a multi-center team from University College London, and elsewhere.
I’m thrilled to add, days after this blog posted, that FDA has just approved Spinraza to treat spinal muscular atrophy, a disease responsible for more infant deaths than any other single-gene condition. Biogen of Cambridge, Massachusetts will be marketing the treatment, which was developed by Ionis Pharmaceuticals of Carlsbad, California, rebranded from the long-standing company Isis Pharmaceuticals about a year ago. I was going to write a separate post about this wonderful success story this week, and then realized I’d already done so. Here’s the link. I love when the biotechnologies born in the 1980s and 1990s conquer the Mendelian diseases, one by one.
BREAKDOWN (not really): CAR-T Therapy
In 2013 and 2015, chimeric antigen receptor (CAR-T) technology saved Emily Whitehead and Layla Richards, from acute lymphoblastic leukemia (ALL). Emily is now well enough to have her own blog, and DNA Science told Layla’s story here.
But in 2016, five patients died in a CAR-T clinical trial. FDA placed the phase 2 clinical trial of JCAR015 in adult patients with relapsed or refractory B cell ALL on hold in July after the first 3 deaths, allowed it to continue, and then shut it down following two more deaths in November.
CAR-T technology is often called a vague “immunotherapy” or “re-engaging the body’s immune system,” with war-inspired metaphors. It’s actually a clever biochemical hybridization of sorts.

A CAR is an engineered receptor that juts from T cells (the “T” in CAR-T). The receptor is not seen in nature, part antibody and part signaling molecule. When the CAR-T cells home to the cancerous B cells in the patient, they evoke an immune response that destroys the cancer cells. The two cases that made headlines used pre-CRISPR methods of gene editing, TALENS for Layla and zinc finger nucleases for Emily.
The five adult patients who died in the Juno Therapeutics “ROCKET” trial, which also deployed CAR-T cells to treat ALL, developed cerebral edema. The culprit seems to be a chemotherapy drug, fludarabine, that had been added to the pre-conditioning regimen to help clear out abnormal cells to make room for the fixed ones – an approach that has helped in other gene therapies and in stem cell transplants using a different cancer drug, busulfan.
So the setback may indicate a need to revamp the pre-procedure pharmaceutical protocol, not retool CAR-T. The road to gene therapy wasn’t without bumps either, such as Jesse Gelsinger’s death from an overwhelming immune response and the boys who developed leukemia when their viral vectors torpedoed oncogenes.
Fortunately CAR-T progress may overshadow the setback. Promising results were recently reported at the American Society of Hematology annual meeting for lymphoma, chronic lymphocytic leukemia, and ALL. The strategy is also being applied to treat ovarian cancer and HIV/AIDS, and other indications are in preclinical stages.
A BRIDGE: FIRST DRUG FOR DMD

FDA’s approval of Exondys 51 (eteplirsen) September 19 is a breakthrough because it’s the first drug to treat Duchenne muscular dystrophy (DMD). Perhaps more importantly, it is a bridge in the sense that patients and their families took an active role in the drug’s approval – generating much controversy.
Eteplirsen is an “exon-skipping” drug. Exons are the parts of genes that are translated into protein, and they are interspersed with introns that are spliced out. Two years ago, a DNA Science post explained how exon-skipping drugs work. The idea is based on a disease, familial dysautonomia (FD), in which some patients have mild symptoms because in some cells, the exon that includes the causative mutation is skipped when the gene is expressed. (This is a Jewish genetic disease, and the phenomenon weirdly reminds me of Passover, when the death-to-the-firstborn mandate skipped over homes marked as Jewish.)
The new DMD drug plunks a synthetic piece of nucleic acid (a morpholino) over the mutation in exon 51 that causes about 13% of cases, enabling production of a stunted but somewhat functional dystrophin protein. Dystrophin makes up less than 0.2% of the muscle proteins but is absolutely critical for the abundant actin and myosin filaments to withstand the force of contraction. Without it, muscles fill with fat.
The approval of eteplirsen was a morass of issues, both technical and regulatory. “Approving a Problematic Muscular Dystrophy Drug: Implications for FDA Policy,” in the December 13 JAMA, by Aaron S. Kesselheim, MD, JD, MPH and Jerry Avorn, MD, provides details.

The clinical trial that led to eteplirsen’s approval tested only 12 patients over 48 weeks, with a double-blind crossover design. The primary endpoint was increase in the amount of dystrophin in skeletal muscle biopsies – which was about 0.9% after 3 years. Results for the standard “6-minute walk test” didn’t improve, until the researchers recalculated, leaving out the two sickest boys. Then the remaining boys did statistically significantly better.
In 2015, FDA rejected a similar drug, drisapersen, following no improvement after 24 weeks and possible adverse effects. That trial had a much larger sample size but didn’t use dystrophin level as the primary endpoint.
FDA’s scientific staff and external advisory committee recommended NOT approving eteplirsen, citing poor performance in the clinical trial. But then the agency held an advisory committee meeting in April and more than 1,000 citizens showed up, mostly affected families or those supporting them, and 4 hours of blisteringly emotional testimony ensued that swept social media and the news. Of course it is emotional – DMD is lethal in the teens or early twenties and without treatment. It almost exclusively affects males because the dystrophin gene is on the X chromosome.
Who would deny desperate parents hope? Might the small but steady production of the precious protein over years rather than months restore some mobility? Extend life? I think it might, and apparently Janet Woodcock, MD, director of the Center for Drug Evaluation and Research at FDA, did too. She overruled the committees to allow approval, with the support of FDA commissioner Robert Califf, MD.
The same day that the JAMA Viewpoint appeared, President Obama signed the 21st Century Cures Act into law. The next day, Dr. Woodcock said on CNBC that one of the things that the act does is “elevate the voice of the patient in how drugs are developed. It enables patients to tell us what matters to them about the disease – what they want to be ameliorated and what is the burden of treatment and problems with current treatment.” She pointed out that the DMD trial was conducted on a “small subset of children with a very serious, life-shortening disease.”
 But … Sarepta Therapeutics cited an annual cost of the drug at $300,000. And that’s the elephant in the room that is perhaps the flip side to Dr. Woodcock’s argument – to what extent will the treatment affect a boy’s quality or length of life, if at all, and at what cost?
But … Sarepta Therapeutics cited an annual cost of the drug at $300,000. And that’s the elephant in the room that is perhaps the flip side to Dr. Woodcock’s argument – to what extent will the treatment affect a boy’s quality or length of life, if at all, and at what cost?
Is eteplirsen’s approval a warning bell? Drs. Kesselheim and Avorn seem to think so. “Patients with DMD need better treatments, and drugs like eteplirsen might one day fill that role. For now, though, the drug has provided a worrisome model for the next generation of molecularly targeted therapies: demonstrate a slight difference in a laboratory test, activate the patient community, win approval, and charge high prices, while relying on limited regulatory follow-up,” they write.
THE BIGGER PICTURE — GENETIC MODIFICATION IN MEDICINE
 My choices for the breakthrough, breakdown, and bridge for 2016 are all genetic modifications modeled after, but not seen in, nature:
My choices for the breakthrough, breakdown, and bridge for 2016 are all genetic modifications modeled after, but not seen in, nature:
• Gene therapy delivers human genes aboard manipulated viruses
• CAR-T treatments combine signaling and antibody parts
• Exon skipping hides a mutation with a synthetic nucleic acid
What constitutes a “breakthrough” or coveted place on a “best of” list is all about context. “Genetic modification” as in “GMO” is a dirty word when applied to crops or to enhance people. Yet it will be the basis of precision medicine in the years to come.
2017? I’d bet on that first gene therapy approval as Breakthrough of the Year — if anyone’s paying attention.