Gene Action Tracks an Autoimmune Disease: Systemic Sclerosis
Systemic sclerosis (Ssc) is a rare disease in which collagen gloms up internal organs and toughens the skin into an armor of sorts. It’s also called scleroderma, from the Greek for “hard” (skleros) and “skin” (derma). Ssc is autoimmune, not inherited, but a recent report in JCI Insight describes how gene expression profiling — transcriptomics – can add precision to diagnosis, monitor response to treatment, and identify drugs that might be repurposed to target SSc.
“The Body Turning to Stone”
 The first known description of SSc as thickened skin is from Hippocrates, circa 400 BC. In 1752, a physician in Naples, Dr. Carlo Curzio, described the case of a 17-year-old:
The first known description of SSc as thickened skin is from Hippocrates, circa 400 BC. In 1752, a physician in Naples, Dr. Carlo Curzio, described the case of a 17-year-old:
“Her complaint was an excessive tension and hardness of her skin over all her body, by which she found herself so bound and straightened that she could hardly move her limbs,” calling her skin “woodlike” and “a dry hide.” She couldn’t open her mouth or fully close her eyes. Another Italian physician, Giovambattista Fantonetti, coined the term “scleroderma” in 1836 to describe a patient with restricted movement from stiff joints and brownish, leathery skin. One patient called her disease “the body turning to stone.”
The name “systemic sclerosis” reflects effects on organs other than skin. Collagen fibers build up in the heart, lungs, gastrointestinal tract, kidneys, nerves, and muscles, amidst sprinklings of tiny cysts. Gummed up joints bend and clench the fingers, while hardening blood vessel linings block circulation to the fingertips and toes, ulcerating them.
Ssc affects only 2.3-10 people per million. The immune system is in overdrive, shooting complex cascades of activated enzymes and cytokines that unleash changes in several cell types: mast cells, macrophages, T cells, and the abundant fibroblasts that pump out the drowning collagen. At the same time, the protein recipes of the extracellular matrix – the gunk surrounding cells, knitting them into tissues – changes. The result is widespread fibrosis – fibers – and sclerosis – hardening.
Enter Transcriptomics
The mantra of molecular biology is that genes are transcribed into RNA, which is translated into protein. Much of genetic testing focuses on identifying mutations — changes in the DNA sequence. But the types and abundances of messenger RNAs (mRNAs or transcripts) offer a subtext that paints a different view of physiology.
All cells have the mRNAs that dictate the “housekeeping” functions necessary to be alive, as well as the mRNAs that sculpt specialized features. But the profile of waxing and waning expression of distinct subsets of genes can veer off course when something is wrong.
 Consider spinal cord injury. A textbook illustration depicts the segment the injury affects; transcriptomics reveals molecular events. A body of literature follows experiments in which researchers injure the spinal cords of rats and then use microarrays to identify the mRNAs produced as the shocked system begins to heal. This approach revealed genes expressed just after the injury whose participation researchers never suspected – the same suite of genes whose protein products heal injury to the deep layer of the skin. The discovery suggested new drug targets.
Consider spinal cord injury. A textbook illustration depicts the segment the injury affects; transcriptomics reveals molecular events. A body of literature follows experiments in which researchers injure the spinal cords of rats and then use microarrays to identify the mRNAs produced as the shocked system begins to heal. This approach revealed genes expressed just after the injury whose participation researchers never suspected – the same suite of genes whose protein products heal injury to the deep layer of the skin. The discovery suggested new drug targets.
More recently, researchers from Spain used gene expression profiling to track the next step in spinal cord injury – treatment. They transplanted bone marrow-derived mesenchymal stromal (stem) cells or olfactory ensheathing cells (glia) from culture into rats just after or a week after injury, then tracked the spectrum of mRNAs produced 2 and 7 days later. Right after injury, the transplanted cells hiked expression of genes whose encoded proteins aid regeneration, but also boosted expression of genes associated with ousting foreign cells from the body – immune rejection.
Beyond rats, tracking changes in gene expression has guided personalization of cancer treatment for years. I wrote about it in 2003 and 2007.
A New Approach to Monitoring Systemic Sclerosis
The new paper from Purvesh Khatri, PhD, from the Stanford Institute for Immunity, Transplantation and Infection, applies transcriptomics to help diagnose, monitor disease progression, and predict response to treatment for Ssc. The FDA has not approved any drugs specifically for the condition, but health care providers prescribe certain immunosuppressants off-label.
The traditional way to diagnose Ssc is rather low-tech – pinching the skin in 17 parts of the body and assigning a value of 0-3 to each, based on thickness. Khatri’s team proposed that patients might share a unique gene expression pattern, and following it might provide a more objective assessment tool.
 They found 415 genes whose expression differs in the condition, with 211 genes overexpressed and 204 underexpressed. Unlike the pinch score, the “SSc Skin Severity Score” (4S) that they developed from the mRNA profile is the same all over a patient’s body, unaltered by jelly bellies and sagging rears, even prevailing against provider-to-provider pinching differences. More importantly, the 4S profile changed to closer to normal 12 months after patients started taking a particular drug, compared to the 24 months required for pinching to indicate a response to treatment.
They found 415 genes whose expression differs in the condition, with 211 genes overexpressed and 204 underexpressed. Unlike the pinch score, the “SSc Skin Severity Score” (4S) that they developed from the mRNA profile is the same all over a patient’s body, unaltered by jelly bellies and sagging rears, even prevailing against provider-to-provider pinching differences. More importantly, the 4S profile changed to closer to normal 12 months after patients started taking a particular drug, compared to the 24 months required for pinching to indicate a response to treatment.
Dr. Khatri and his colleagues are very excited about one of the implicated genes, which encodes the epidermal growth factor receptor (EGFR) — several genes in the systemic sclerosis “signature” encode proteins that bind the receptor. It makes sense, because EGF partakes in extracellular matrix deposition and regulates the division of skin fibroblasts. And people with Ssc have autoantibodies to EGFR.
The implication of EGFR is good news, because several EGFR inhibitors are on the market to treat cancer. Dr. Khatri explains the hypothesis: “the immune system is trying to get rid of cancer cells, as it should. However, in the process it starts to recognize the normal cells as antigens and attacks them, causing in autoimmune disorder. Our paper, and unpublished experimental data, suggest that there is a potential for EGFR targeting FDA-approved drugs to treat systemic sclerosis patients, such as Geftinib. Fingers crossed!”
A WORD ON “THE MAN IN THE HIGH CASTLE”, SEASON 2
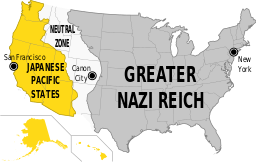 I’ve just finished watching season 2 of Amazon video’s terrifying The Man in the High Castle. It depicts an alternate reality in which Germany and Japan won the second world war, with an atomic bomb taking out Washington, DC — as well as other possible fates. I admit to some confusion over plot holes and teasers — that’s how they hook you into watching the next season — but I did catch enough of the dialog to suspect that genetic info about one of the families is wrong.
I’ve just finished watching season 2 of Amazon video’s terrifying The Man in the High Castle. It depicts an alternate reality in which Germany and Japan won the second world war, with an atomic bomb taking out Washington, DC — as well as other possible fates. I admit to some confusion over plot holes and teasers — that’s how they hook you into watching the next season — but I did catch enough of the dialog to suspect that genetic info about one of the families is wrong.
Thomas Smith, adolescent son of Obergruppenführer John Smith, has a form of muscular dystrophy. According to a footnote in Wikipedia, it is Landouzy-Dejerine syndrome, which I hadn’t heard of by that name but recognized its name today, facioscapulohumeral muscular dystrophy (FSHD). It’s autosomal dominant – if you have it, so does or did one of your parents, unless you’re a new mutation (about 30 percent of cases).
In a 10-second scene in the final episode, we learn that John Smith’s uncle had the disease, and that his two daughters (Thomas’s sisters) “must be” carriers.
Well, no.
A dominant condition doesn’t have carriers. A person has the mutation or doesn’t, and if symptoms don’t appear until later in life, she or he is pre-manifest, not a carrier. The fact that Thomas’s uncle had it, and was presumably murdered for the imperfection, as well as the erroneous information that his sisters are carriers, indicates that in this family it is inherited, not due to a new mutation.
If the inheritance pattern is autosomal recessive, then John’s family would have to carry it as well as his wife’s — unusual for a disease this rare. I can only reconcile these discordant genetic facts if John Smith has the mutation but is non-penetrant – meaning he has the genotype but not the phenotype. I wonder what the Nazis would do with that intel if they’re so interested in sanitizing the gene pool? Plus, Thomas has seizures and he trips – not the upper body weakness of the disease.
I suppose the writers chose this form of muscular dystrophy because it isn’t as horrible as others like Duchenne, making it more horrible that the Nazis exterminate those who have it. When Thomas realizes that he’s not healthy, he mutters “I’m a useless eater” and “I’m defective.”
I won’t spoil the ending. But can anyone explain an inheritance pattern that has carriers and non-penetrance? Maybe the new showrunner for season 3 will figure it out while the rest of us try to understand the end of season 2.
