Blinded By Stem Cells
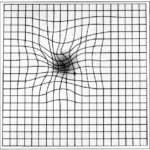
The small wavy shimmers and fuzzy areas in the 78-year-old’s eyes had grown slowly, leading to a diagnosis of age-related macular degeneration (AMD), which affects more than 10 million Americans. She had the more common
“dry” form. Despite 2 years of injections of one of the latest drugs (a vascular endothelial growth factor [VEGF] blocker), her sight was worsening. How much longer would she be able to drive, with a growing blob obscuring the center of her visual world?
Her family went online to research alternative approaches.
“They found a clinic in south Florida offering stem cell injections to treat macular degeneration and the clinic advertised that they were conducting a trial. They had a protocol listed on ClinicalTrials.gov,” recalled Thomas Albini, MD, an ophthalmologist at the Bascom Palmer Eye Institute of the University of Miami, who treated the woman. An article in this week’s New England Journal of Medicine describes that case and two others, all older women who lost their vision following stem cell treatment for AMD at the same clinic in south Florida, in June 2015. For details see my write-up in Medscape Medical News.
The website of the stem cell company that supplied the unnamed clinic is rather sparse in detail now, perhaps because of a settled civil lawsuit and the NEJM paper. But back in spring 2015, it mentioned entry NCT02024269 at ClinicalTrials.gov, a “Study to Assess the Safety and Effects of Cells Injected Intravitreal in Dry Macular Degeneration.” The “Official Title” looked legit too, despite the botched grammar: “An Open-label, Non-Randomized, Multi-Center Study to Assess the Safety and Effects of Autologous Adipose-Derived Stromal Cells Injected Intravitreal in Dry Macular Degeneration.”
Translation: Stem cells from fat. A patient’s own.
I don’t know what the entry for NCT02024269 looked like when the patients saw it, but now, stamped at the top in red, is: This study has been withdrawn prior to enrollment.
Next to Enrollment: 0. A big fat zero.
 Buried in the details remains the requirement that participants be legally blind, which five of the six eyes of the three women weren’t. The patients each paid $5000 for the procedure, which was administered into both eyes at once. According to a talk that Dr. Albini gave to the FDA in September 2016, a nurse practitioner injected the cells.
Buried in the details remains the requirement that participants be legally blind, which five of the six eyes of the three women weren’t. The patients each paid $5000 for the procedure, which was administered into both eyes at once. According to a talk that Dr. Albini gave to the FDA in September 2016, a nurse practitioner injected the cells.
The clues – patients paying, no patients in a withdrawn trial, and not being visually impaired enough to meet inclusion criteria – indicate that the women had not actually been in a clinical trial. A spokesperson for the company confirmed to me that they hadn’t been enrolled in a clinical trial, but they had indeed received the company’s product. In Kellyanne fashion, she repeated odd phrases that didn’t reveal much, for legal reasons.
A TALE OF THREE WOMEN
According to the NEJM report, at least two of the women and possibly all three thought they were in a bonafide clinical trial. It would’ve been easy to connect the dots from the company website to ClinicalTrials.gov to the treatment. And they did sign consent forms.
Whether the women were or were not registered in an FDA-regulated clinical trial, they were clearly in trouble.
Two days following the procedure, her eyes painful and bleeding, the first woman went to the Center for Sight in Sarasota and was quickly referred to the Bascom Palmer Eye Institute, where Dr. Albini and colleagues saw her. She was legally blind, although she’d started out at 20/50 in the right eye and 20/100 in the left eye.

Patients with very low visual acuity move from the familiar 20/20 Snellen eye chart to a more qualitative scale of what they can see: “counting fingers,” “hand motion,” “light perception,” and “no light perception.” The first patient seen could count fingers only, quite different from her pre-procedure concern about failing central vision affecting her driving. Optical coherence tomography, which images the retina in cross section so that it looks like one of those little Italian pastries with the colored layers, showed swelling and thickening of the central retina in the right eye, and bare areas like a desert in the left. Within two months, her retinas had detached. They were reattached and she regained some vision, but remains legally blind.
The second patient to come to the researchers’ attention, just a few days later, was more severely injured. She was left with “no light perception” in both eyes; she’d started out with 20/60 in the right eye and 20/30 in the left. She too had found the company website, which had lead to NCT02024269, and paid $5,000.
The researchers contacted the American Society of Retina Specialists, which alerted its members, and soon a third patient turned up. She’d flown from Oklahoma to Florida for the procedure. She was 88, had appeared at the clinic with 20/40 vision in her right eye and 20/200 in the left, and came to Bascomb with just hand waving in her right eye and light perception in her left.
TRUMP’S ToFOO
The silver lining to the tragic tale is that the NEJM report might strengthen FDA’s efforts, begun three months after the women were treated, to regulate stem cell therapies. But I’m concerned about repercussions of the January 30 Presidential Executive Order on “Reducing Regulation and Controlling Regulatory Costs.” The seemingly arbitrary declaration that “any new incremental costs associated with new regulations shall, to the extent permitted by law, be offset by the elimination of existing costs associated with at least two prior regulations,” immediately dubbed the “Two-For-One Order” or ToFOO, is expected to dampen efforts to institute any new regulations – perhaps even ones so desperately needed as efforts to curb stem cell tourism.
 Our articulate leader provided less complex alternative language in a talk to pharma execs: “When you have a drug, you can actually get it approved if it works, instead of waiting for many, many years. We’re going to be cutting regulations at a level that nobody’s ever seen before, and we’re going to have tremendous protection for the people.” It’s gonna be great, I’m sure.
Our articulate leader provided less complex alternative language in a talk to pharma execs: “When you have a drug, you can actually get it approved if it works, instead of waiting for many, many years. We’re going to be cutting regulations at a level that nobody’s ever seen before, and we’re going to have tremendous protection for the people.” It’s gonna be great, I’m sure.
Judging by the viewpoints of the recommended new FDA commissioner Scott Gottlieb, MD about how clinical trials for treatments for certain rare genetic diseases might have been more humanely designed and conducted, I’m hopeful that he’ll champion regulation of stem cell therapies as a drug, despite the inanity of ToFOO.
STEM CELL REGS
FDA’s scrutiny of a second stem cell company intensified right about when NCT02024269 posted its final update.
The agency began a correspondence in mid September 2015 with the company, which had facilities in Florida, California, and New York. Their product, also based on fat stem cells, targeted a kitchen sink of maladies, including autism, Parkinson’s disease, chronic obstructive pulmonary disease, multiple sclerosis, cerebral palsy, amyotrophic lateral sclerosis, and AMD. FDA, in the first letter of the volley, insisted that stem cells used in treatment are drugs and should be regulated as such.
The company that the letter targeted claims to have halted its use of “stromal vascular fraction (SVF) product” (the fat stuff) September 20, 2015. But FDA sent a harsh and detailed warning letter, dated December 30, 2015, reiterating that the product must be regulated as a drug because it is intended to treat, and is also a biological and a human-cell-based product.
Use of autologous (“from self”) stem cells dates to an obscure regulation, the Public Health Services Act of 1944, that describes skin and vein grafts. The “cells out, cells in,” approach that the company that treated the three blind women uses, according to their rep (but not on eyes anymore) isn’t experimental and doesn’t require FDA approval. The company released this statement just before the NEJM published: “Founded in 1999, U.S Stem Cell, Inc. has been committed to the research and development of effective cell technologies to treat patients with a variety of diseases and injuries. Since 2001, our clinics have successfully conducted more than 7,000 stem cell procedures with less than 0.01% adverse reactions reported.”
Injecting a bloblet of fat stem cells into an eyeball isn’t quite the same as a skin or vein graft, which is same-to-same tissue. And the more a product is tweaked or veers from normal biology, even if it originates in the patient’s body, the stricter the regulatory requirements. That means filing an Investigational New Drug Application and conducting a real clinical trial, with countable participants and experimental controls to assess safety and possibly efficacy. ”Lots of people are giving these therapies without studying them and without any efficacy and safety documentation,” Dr. Albini said.
 I can’t fathom why people – practitioners and patients — don’t seem to regard injecting stem cells with the same caution that they do gene therapy. My book The Forever Fix tells the story of gene therapy for a retinal disease that also injects a biological product beneath the retina. The clinical trial began in 2008 and has recently completed phase 3, from Spark Therapeutics. That trial, and in fact eye procedures in general, are done one eye at a time, so that a disaster won’t blind the patient. It’s common sense and common practice.
I can’t fathom why people – practitioners and patients — don’t seem to regard injecting stem cells with the same caution that they do gene therapy. My book The Forever Fix tells the story of gene therapy for a retinal disease that also injects a biological product beneath the retina. The clinical trial began in 2008 and has recently completed phase 3, from Spark Therapeutics. That trial, and in fact eye procedures in general, are done one eye at a time, so that a disaster won’t blind the patient. It’s common sense and common practice.
Scared people desperate for treatment may fall for the white coats and official language of websites for companies offering stem cell treatments, which continue to proliferate. A study from August 2016 found stem cell procedures provided at 570 clinics in the US, representing 351 companies.
IN DEFENSE OF FAT

Stem cells from fat have been pitched to the public for years. Regenerative Orthopedics Institute, for example, supplies stem cells to treat osteoarthritis, sports injuries, and overuse syndromes. Adipose tissue is a rich repository of the healing cells, better even than bone marrow, and certainly easier to part with thanks to liposuction.
The veterinary market is robust. Arthritic dogs and lame horses have enjoyed restored function following injection of their own stromal vascular fraction in a soup of platelet-rich plasma that’s rich in growth factors.
 VetStem Biopharma’s website offers wonderful testimonials on the healing abilities of fat stem cells, such as Honey the therapy dog who ruptured ligaments in her knees and shoulders and Smokey the horse, who severed a tendon in a leg.
VetStem Biopharma’s website offers wonderful testimonials on the healing abilities of fat stem cells, such as Honey the therapy dog who ruptured ligaments in her knees and shoulders and Smokey the horse, who severed a tendon in a leg.
But human eyes are a different matter. Reports on treating eye diseases with adipose-derived stem cells are few. In the first report, to treat retinitis pigmentosa, the researchers conclude that the procedure “should be applied with caution” due to eye-related complications in 6 of their 11 patients.
A PATIENT CHECKLIST
While stem cell tourism remains alive and well, I hope that this week’s report in the top medical journal will make people stop and think before they submit to an untested therapy.
Although Dr. Albini calls the plight of the three women a worst-case scenario, I can imagine even more devastating places in the body to introduce stem cells, life-threatening ones. And of course stem cells, being stem cells, in addition to what the media often simplistically call “turning into” specialized cells, continually spin off more and more stem cells – which, if uncontrolled, could seed a cancerous growth.
 Sums up Timothy Caulfield, research director of the Health Law Institute at the University of Alberta, and co-author of the August 2016 survey of stem cell companies, “This is a tragic example of how the premature use of an unproven stem cell intervention can go wrong. Though a very promising field and despite all the hype, very few stem cell therapies have been shown to be effective and safe.”
Sums up Timothy Caulfield, research director of the Health Law Institute at the University of Alberta, and co-author of the August 2016 survey of stem cell companies, “This is a tragic example of how the premature use of an unproven stem cell intervention can go wrong. Though a very promising field and despite all the hype, very few stem cell therapies have been shown to be effective and safe.”
The disaster of the three women has been graciously called a misunderstanding, and not outright deception, but I can’t help but wonder about the role of the entries at ClinicalTrials.gov. The website has valuable information, resources, and contacts, but does the “gov” at the end of the link suggest validity? Do health care consumers automatically trust an official-seeming government website aimed at researchers? Unfortunately clicking through to the Disclaimers leads to general statements about NIH web material. Are entries actually scrutinized? About all I do know is that “observational” studies, such as on a large population, do not require FDA oversight. But the NCT02024269 trial that the patients thought they were in was “interventional.”
So with that caveat, as well as the fact that the informed consent process is indeed rigorous for real clinical trials in the US, here’s a checklist for patients to help determine whether an entry at ClinicalTrials.gov is operating under an Investigational New Drug application from FDA, where stem cell therapies fall under Cellular and Gene Therapy Products:
• A participant in a clinical trial should not be paid or asked to pay.
• The design description should indicate specifics: how many years will the study last? Will participation require travel, and if so how often and for what duration?
• Are eligibility criteria, like age and sex, met?
• Are inclusion criteria met, such as legal blindness?
• How is the treatment delivered? Pill? Injection? Infusion? Surgery?
• How many drugs and procedures in addition to the treatment are part of the protocol?
• Are the ethics acceptable, such as randomization for placebo?
• Has the trial been delayed? Check “first received” against updates.
• Read and heed warnings, such as “Participants’ safety is not guaranteed.”
Until better (or any) regulations are in place for stem cell treatments, caveat emptor! Let the buyer beware.
