What’s in a Placenta? A Transcriptomic View
I didn’t think much about placentas until I had one and part of it dislodged halfway through a 4-mile run. Happily it ended well some months later with a “small for gestational age” tiny baby and a “large for just giving birth” me.
After that I became more sensitive to mentions of this intriguing organ that connects two individuals in a way that no other does. I noticed new hair conditioners touting “placenta” as an ingredient, and learned about people eating them. A geneticist friend kept one from a daughter’s birth, pickled in a jar, in his lab.
A placenta is much more than an “afterbirth,” a biological afterthought. It’s crucial.
PLACENTAL MAMMALS RULE
A human placenta may appear to be nothing more than a bloody blob that swooshes out in the wake of a baby, but it nurtures the members of 4400 or so species while they develop inside their mothers. That’s preferable to hauling oneself, helpless and blind, out of a pouch and climbing along fur to a glorified sweat gland trying to be a nipple, like marsupials such as kangaroos and opossums do. The third type of mammal, a monotreme, hatches out of an egg. No wonder the placental mammals have taken over, and range today from tiny shrews to gigantic blue whales.
And so when I saw an article probing the “cell communication network of the maternal-fetal interface” in this month’s Genome Research, I was intrigued. Not only did it attempt to answer questions that I’ve long had, but it catapulted me back to graduate school at Indiana University, where I was lucky enough to have taken the first offering of a course, “Embryos and Ancestors,” from Rudy Raff and Thom Kaufman. From that course sprung the field of evolutionary developmental biology, aka “evo-devo.”
Back then, we talked about vague “gradients of morphogens” guiding development of an embryo. Technology has brought precision. The new work pinpoints the genes behind one spectacularly important process for most of us mammals: formation of the placenta.
PEEKING INTO SINGLE CELLS
 Mihaela Pavličev, PhD of the Center for Prevention of Preterm Birth, Perinatal Institute, Cincinnati Children’s Hospital Medical Center and colleagues use the word “negotiation” to describe the unique relationship that must be established early in a pregnancy and then maintained, for a birth to result.
Mihaela Pavličev, PhD of the Center for Prevention of Preterm Birth, Perinatal Institute, Cincinnati Children’s Hospital Medical Center and colleagues use the word “negotiation” to describe the unique relationship that must be established early in a pregnancy and then maintained, for a birth to result.
“We cannot understand pregnancy by focusing on the fetal side (placenta) alone, or on the maternal (uterus) alone. How do we maintain stability, not as a war, but rather without damage to mom or fetus? Most pregnancy defects can be seen as interrupting this temporary stable unit. But to be able to study that, we need to know which cells are talking to which other cells,” explains Dr. Pavličev.
Instead of looking macroscopically at the pancake-like organ, they conducted an elegant and extensive analysis of individual cells, using single-cell transcriptomics. (I’m departing from not calling a pregnant woman a mother because when discussing a placenta, it’s important to name the sides: fetal and maternal.)
“Transcriptomics” catalogs and quantifies the messenger RNA transcripts in a cell, which glimpses the crescendos of gene activity in a particular situation. Understanding the fetal-placental crosstalk may have clinical applications, from a test of menstrual matter to predict risk of early miscarriage to understanding pregnancy and birth complications that stem from placental dysfunction.
HOW A PLACENTA FORMS
A lone sperm leading a pack of about 200 approaches an egg and then burrows through the protective layers, sending its genetic package through as its middle piece and waving tail fall away. Inside the giant cell, pronuclei meet and merge as fertilization (conception) completes.
Over the next few days, cleavage divisions ensue as 2 cells become 4, 4 become 8, and so on, forming a solid ball of cells that then hollows out. Now a blastocyst, the early embryo approaches a uterine (aka Fallopian) tube.
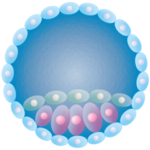
Soon, a smidgeon of cells collects on the interior. This inner cell mass (ICM) will develop into the embryo and some of its baggage (the yolk sac, amnion, and allantois). The other cells (trophoblasts) that form the boundaries of the ball (trophectoderm), however, are not just there for the ride, like a hamster ball – they will become the placenta.
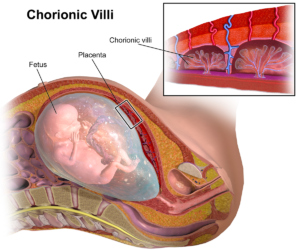
Once in the uterus, the tiny ball of cells lands and settles into the uterine lining, into which the trophectoderm tentatively extends finger-like projections, appropriately called chorionic villi. Meanwhile, the lining has switched from relentless cell division mode to becoming more glandular, rich with blood and nutrients, and called the decidua. Like deciduous trees so-named because they shed their leaves, the decidua is shed as menstrual flow if fertilization doesn’t happened.
But if an embryo burrows in, the uterine lining develops an intricate bed of blood vessels to welcome the chorionic villi that dip into the forming pools of maternal blood. Oxygen and nutrients will transfer from the blood pools across the villi to the embryo and fetus, as wastes such as carbon dioxide move in the opposite direction. All is good.
 The chorionic villi develop into the placenta, the job complete about when embryo becomes fetus, meaning rudiments of all major structures have formed. And like in other organs, not all cells that make up the placenta specialize in the same way. Standard histological methods can miss rare cell types. That’s where single-cell transcriptomics comes in.
The chorionic villi develop into the placenta, the job complete about when embryo becomes fetus, meaning rudiments of all major structures have formed. And like in other organs, not all cells that make up the placenta specialize in the same way. Standard histological methods can miss rare cell types. That’s where single-cell transcriptomics comes in.
Some placental cell specializations are obvious: cells that form the outside of chorionic villi do different things than the cells on the inside. Most important is the actual interface between embryo/fetus and mother, called the syncytiotrophoblast. (A syncytium is a cell with several nuclei, usually pretty big, like a skeletal muscle cell.)
The researchers started with two placentas collected at births and interrogated 87 single-cell transcriptomes. The cells fell into 5 groups based on which genes were turned on or off: two groups of 28 cells each, one of 23 cells, and two smaller groups, one of 5 cells and one of 3. Only the 3-cell group was maternal. They added a few control collections, like cells shed in a menstrual flow and other syncytiotrophoblasts.
The three big groups came from inside chorionic villi, and the 5-celled group from outside, showing that the difference in location is more than meets the eye – gene expression differs even within the same part of a part.
Quite a lot goes on in a placenta. Here’s what the tallying of the cell functions revealed:
• As a placenta forms, cells destined to specialize as the same type turn on or off sets of genes in different orders. This is protective, like cars taking different routes to the same destination – chances are at least some will get there.
• Members of the 5-cell group are “extravillous trophoblasts,” or EVTs. They invade the uterine lining, build the blood supply (by sporting receptors for vascular endothelial growth factor [VEGF] and knitting endothelial cells into tubules), and then shield the embryo from the maternal immune system.

• Syncytiotrophoblasts, the special interface cells, crank out the hormones and glycoproteins that keep the pregnancy going, as well as several HLA proteins that are also seen in the powerful dendritic cells of the immune system.
• The 3 cells of the smallest group, the maternal ones, act like antigen-presenting cells, accepting the fetus rather than ousting it as if it was a growing parasite or mismatched transplanted organ.
Comparing the shreds of uterine lining in a menstrual flow to the lining during pregnancy confirmed the profound change from proliferative to decidual, which dampens division and hikes production of hormones, metabolic molecules, and immune system components.
HIGHLIGHTING CELL-CELL CHOREOGRAPHY
Next the researchers deduced cell-cell interactions by looking for the hallmarks of cellular communication: telltale transcripts encoding receptor proteins and the molecules – ligands – that bind them, a little like a cell phone (the ligand) held to an ear (the receptor). “Gene expression of ligands and receptors alone doesn’t prove crosstalk, but it offers a solid and high throughput starting point,” Dr. Pavličev says.
When chorionic villi reach out towards uterine decidua, they establish a “hub of feto-maternal cell signaling.” The researchers summarized the voluminous data in a colorful and complex diagram that reveals the patterns of gene expression chatter between pairs of decidual cells, dendritic cells, EVTs, and syncytiotrophoblasts. Oddly, the resulting diagram looks a little like the inner cell mass nestled inside its trophoblast. I don’t usually run large images but this one is amazing:
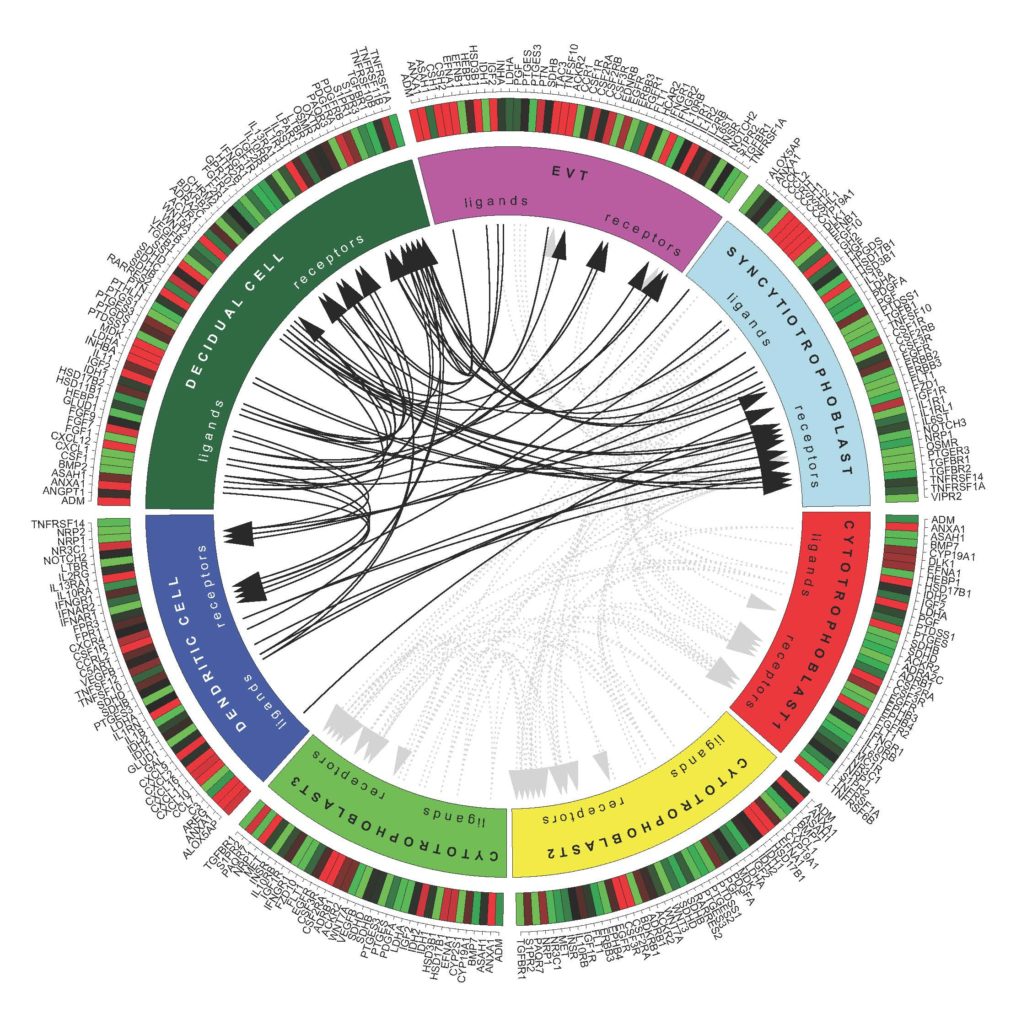
The cells are functionally connected. The decidual cells crank out growth factors for which syncytiotrophoblasts have receptors, as well as immune system proteins (an interleukin and a colony stimulating factor) that bind receptors on the EVTs and dendritic cells.
The intricate paired actions set up a steady-state of sorts, a subtle homeostasis that maintains levels of growth factors and hormones while holding the maternal immune system at bay. Anti-inflammatory molecules counter the effects of inflammatory molecules. “We interpret this as a way to keep stability. Similar to a temperature regulatory circuit, one needs to have the ability to cool or heat present simultaneously,” Dr. Pavličev explains.

In addition to growth, creating a blood supply, and establishing a peaceful co-existence with the maternal immune system is the exquisite hormonal control that guides pregnancy. The RNA analysis confirmed that all types of trophoblast cells secrete progesterone – the hormone given to prevent some miscarriages by maintaining the uterine lining – and that the decidual side secretes estrogens as well as progesterone.
I hope that the new description of placental physiology provides tools to prevent pregnancy loss due to something amiss in the support system. A bigger picture is one of evolution. The finding that placental cells take slightly different routes to specialization is a compelling example of natural selection of a highly adaptive trait.
I am proud to be a placental mammal!