Drug Duo Treats ALS – On A Chip
When a disease is as relentless as amyotrophic lateral sclerosis (ALS; aka Lou Gehrig’s disease; aka motor neuron disease), any promising research result is welcome news.
A study just published in Science Advances shows that two drugs already FDA-approved for other diseases, when teamed, halt neuron death and bolster muscle contraction in an “organ-on-a-chip” model of ALS. When the drugs meet in a device that places tiny balls of motor neurons from a patient next to strips of healthy skeletal muscle, the set-up not only recapitulates the disease, but shows the synergy of the drugs. They are rapamycin (Sirolimus) and bosutinib (Bosulif).
A Slow Decline
ALS gradually destroys motor neurons in the spinal cord, the motor cortex, and the brainstem. Overexcited nerve cells eventually die, withdrawing their stimulation of muscle cells. Movement ceases and the disease reaches the breathing muscles typically within 3 to 5 years. The patient can then choose mechanical ventilation or to let nature take its course.
Early symptoms may be overlooked for a time: difficulty holding a pen or grasping a cup or key, tripping, numb feet, slurred speech, change in vocal pitch, weakness, and fatigue. Muscle twitches that look like snakes squirming under the skin may be the symptom that initially sends a patient to the doctor. Paralysis and muscle atrophy usually spread slowly enough to provide time to adjust households and plan for aids, like devices to assist mobility, eating, and breathing.

I learned about the progression of ALS as a hospice volunteer and as a fan of folksinger Eric Lowen, who continued performing even when his partner Dan Navarro had to carry him onto the stage and then maneuver his wheelchair. (They wrote “We Belong,” the Pat Benatar hit, and many other great songs). Many books describe living with the disease, both fiction and non-fiction. A poignant realistic depiction is in the recent novel Every Note Played, by neurologist Lisa Genova.
The main character, Richard, lives in Boston and is “trapped inside his condo, his living tomb,” increasingly unable to walk but not ready to give up.
One day, after his morning caregiver Bill leaves, Richard sets off for the Public Garden three blocks away. In his pocket is a cell phone programmed with the numbers of neighbors who’ve agreed to help, calling voice-activated. He makes it to a bench, just barely. Then he’s in trouble.
“Three blocks from the front step where Bill left him, he’s completely sapped. His legs have become sandbags, his bones filled with rocks, impossibly heavy, and he lacks the energy to move them.” Passersby misunderstand, refuse to help. Richard waits, rests, and then manages to walk back. Strength gone, he desperately talks to his phone, and although his speech is garbled, he calls a neighbor to ask her to unlock the door. But she doesn’t answer, and Richard has to urgently use the bathroom. It’s a turning point in the book.
From 12,000 to 15,000 people in the U.S. have ALS. Only about 10 percent of cases are inherited. Because knowing a responsible gene reveals a mechanism, many of the experiments that have inspired clinical trials have been conducted on mice with mutations in the superoxide dismutase (SOD1) gene, the most common inherited cause. The fact that most cases are sporadic, not inherited, may partly explain the poor history of drug development for ALS (see “ALS Clinical Trials Review: 20 Years of Failure. Are We Any Closer to Registering a New Treatment?”) Another reason: neuromuscular junctions in mice aren’t exactly like ours.
The only drugs approved for ALS, riluzole (Rilutek) and edaravone (Radicava), have only moderate effects and don’t alter the disease course. Scams and misinformation are rampant.
Despite the high drug failure rate, the pipeline for ALS treatments is impressive, although some entries are restricted to patients with SOD1 mutations. I perused the first 100 of 449 current entries at ClinicalTrials.gov.
The list includes the usual biotech suspects: stem cells, gene therapy, monoclonal antibodies, small molecules with various signal-disruptive or neuron-bolstering effects, exercises, vitamins and food supplements, and devices to stimulate the spinal cord.

Candidates for repurposing include lithium, tamoxifen, antiretrovirals, a seizure med (perampanel), the new spinal muscular atrophy drug Spinraza, MS drug Gilenya, and a drug used to treat heart arrhythmias in cats and dogs, Mexiletine.
Failures include glutamate-tamer Talampanel, neurite-extender Ozanezumab, “novel immune regulator” NP001, cell death blocker TCH346, gene editing treatment SB509, and the antisense drug BIIB067/.
New potential drugs inhibit key enzymes that send signals: GDC-0134, fasudil, and “oxygenated nanobubbles” of RNS60. MN-166 is a pill that reduces inflammation in the brain and spinal cord. It is about to begin a phase 3 clinical trial, and is used in Japan to treat asthma and complications of stroke.
The clinical trials roster may soon include the two drugs that complement on the microfluidic device used in the new study.
ALS-On-A-Chip
Tatsuya Osaki, Sebastien Uzel, and Roger Kamm, of the Department of Mechanical Engineering at the Massachusetts Institute of Technology, invented the fluid-filled tiny silicone chip grooved on the bottom.
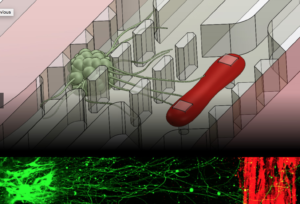
The microfluidic device models a neuromuscular junction, with motor neuron clusters (green) extending axons towards skeletal muscle (red). The fluorescence image below shows the motor neurons sending axons towards the muscle strip over a distance of a millimeter. (Credit: Sebastien Uzel, MIT)
In the reported experiments, one chamber held balls of motor neurons from a sporadic ALS patient, cultured from induced pluripotent stem (iPS) cells made from skin fibroblasts. The other chamber held healthy skeletal muscle cells, also made from iPs cells. Using stem cells is a way to mass-produce cell types that don’t divide and can’t be sampled from a patient.
The motor neurons were engineered to include a light-sensitive gene (channelrhodopsin-2) so that when a newly-innervated muscle contracts, light flashes. Sprinkled amongst the motor neurons were astrocytes, which are star-shaped cells that are also part of neural tissue. They soak up the excess glutamate that over-excites ALS motor neurons.
Like the Jets and the Sharks at opposite sides of the playground in West Side Story cautiously approaching one another, the ALS motor neurons extended axon ends – neurites – through the neutral zone, towards the cylinders of skeletal muscle. Tiny bubbles bud off of each motor neuron, releasing the neurotransmitter acetylcholine into the synaptic gap to reach the nearest skeletal muscle cell.
In control experiments using healthy motor neurons, the ensuing muscle contraction was robust. But with motor neurons from the ALS patient, fewer muscle fibers contracted and those that did grew weaker. The motor neurons degenerated, mostly due to buildup of a protein called TDP-43, and eventually died.
Enter the two drugs.
Rapamycin (Sirolimus) is approved as an immune suppressant to prevent rejection of a transplanted kidney. It belongs to the class of drugs called MTOR inhibitors. Bosutinib (Bosulif) is approved to treat chronic myelogenous leukemia. It’s a tyrosine kinase inhibitor. Both drugs trigger autophagy (“self-eating”), which is how cells clean up accumulated stuff, like the gummy TDP-43 proteins. Because the drugs work in different ways, perhaps together they’d ramp up autophagy.
But in the body, drugs would have to cross the blood-brain barrier and the blood-spinal-cord barrier. To simulate this, the researchers placed a layer of tile-like endothelial cells atop the microfluidic device, which hugs it like plastic wrap melting over leftovers in the microwave. When both drugs were applied, muscle contraction strengthened and motor neurons survived!
The researchers can’t yet explain this “most remarkable finding,” but they suggest how it might happen.
Either drug can’t, by itself, cross the endothelial lining. But the chemistry suggests that when both are applied, bosutinib can settle into the nooks and crannies that form in the endothelial plastic wrap. That activates a protein “pump” in the layer, but with both drugs present it’s inefficient. A bit of bosutinib sneaks through and the other drug latches onto it and enters too, like getting into a show by hanging on to a friend who has a ticket.
And so the tiny microfluidic device, the ALS-on-a-chip, peeked at a dual assault on the cellular setting of a terrible disease. Perhaps clinical trials can now investigate the drug combination – or both drugs prescribed off-label to desperate patients.